Slime is one of the most popular toys for kids, which provides them hours of fun. There are also lots of adults enjoying slime as a stress reliever. Let’s take a look at what chemicals slime is made of.
Slime is a type of polymer, which is a large molecule composed of many smaller molecules, called monomers. It is a product of chemical reaction between two main ingredients; polyvinyl alcohol and tetrahydroxyborate (Borax).
Polyvinyl alcohol is a water-soluble polymer, which is formed by the polymerization of vinyl acetate monomers. Polyvinyl Alcohol is also known as PVA. PVA has many applications in pharmaceuticals, cosmetics, textiles, and food-packaging, as it is nontoxic and noncarcinogenic. Polyvinyl alcohol is also present as individual long polymer chains in PVA glue.
Borax is a naturally occurring alkaline compound. It is also known as sodium borate, sodium tetraborate, or disodium tetraborate. Borax is a white powder that has been used in cleaning products for years, to help get rid of stains and dirt. Borax is also used in cosmetic products as emulsifier or preservative. When borax is dissolved in water, it forms the borate ion.
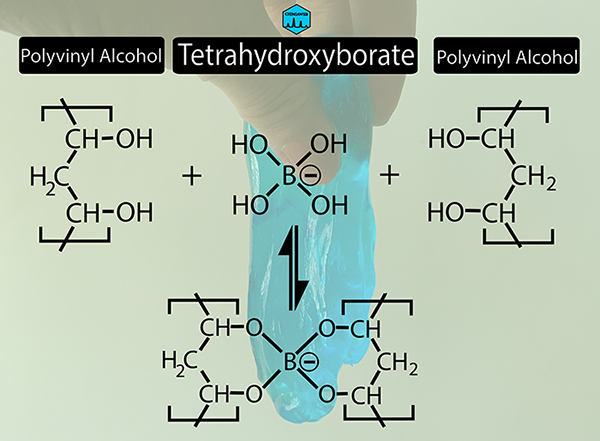
When PVA glue is mixed with borax solution, borate ions cross-link between polyvinyl alcohol chains. This reaction makes the polymer chains not to flow easily as before. Instead, the new created network has more viscosity and elasticity.
The viscosity of slime changes under physical force. Fluids, which show this behavior, are called non-Newtonian fluids. So, slime is considered as a non-Newtonian fluid. In other words, if you apply force to slime, like shaking or squeezing, it cause them to act rigid like a solid. The result of this stress is the formation of cross-links between the chains that increase the viscosity. But when you let it slowly move through your fingers, the viscosity decreases and it flows runnier.

